|
The normal usage in the field is to collectively refer to both the scattering process and the maps of directions as electron diffraction, not differentiating the two. However, it is often also used to denote, collectively, different methods of collecting data on the directions of electrons leaving the sample, what are better called electron diffraction patterns. Strictly, the term electron diffraction refers to how electrons are scattered by atoms, a process that is mathematically modelled by solving forms of Schrödinger equation. Often there will be an array of spots (preferred directions) as in Figure 1 and the other figures shown later.įigure 2: Young's double slit experiment, showing the wave in blue and the two slits in yellow the other Figure with purple waves is similar from a small array of white atoms. A map of the directions of the electron waves leaving the sample will show high intensity (white) for favored directions, such as the three prominent ones in the Young's slits experiment of Figure 2, while the other directions will be low intensity (dark). 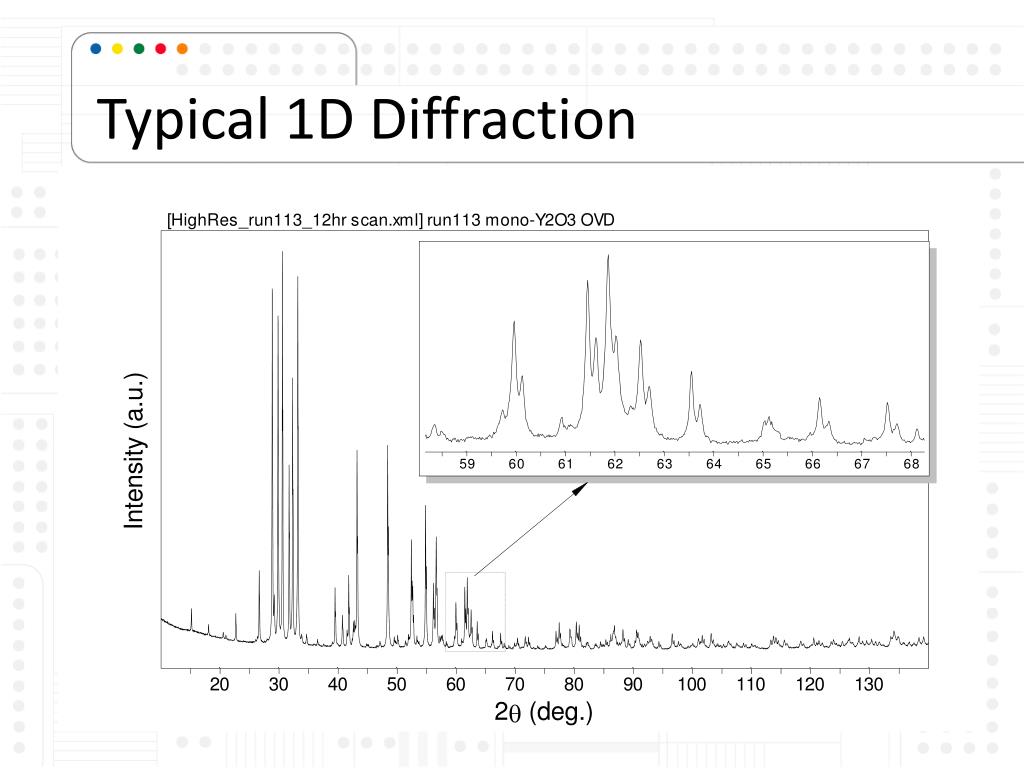
This has relevance for imaging within electron microscopes, whereas electron diffraction patterns are measured far from the sample, which is described as far-field or Fraunhofer diffraction. Ĭlose to the aperture or atoms, often called the "sample", the electron wave would be described in terms of near field or Fresnel diffraction. This is comparable to diffraction of an electron wave where the small dots would be atoms, see also note. If instead of two slits there are a number of small points then similar phenomena can occur as shown in the second image where the wave is coming in from the bottom right corner. After going through the slits there are directions where the wave is stronger, ones where it is weaker – the wave has been diffracted. Waves can move around objects and create interference patterns, and a classic example is the Young's two-slit experiment shown in Figure 2, where a wave impinges upon two slits in the first of the two images. In comparison, x-rays are scattered after interactions with the electron density while neutrons are scattered by the atomic nuclei through the strong nuclear force.Īll matter can be thought of as matter waves, from small particles such as electrons up to macroscopic objects – although it is impossible to measure any of the "wave-like" behavior of macroscopic objects. The negatively charged electrons are scattered due to Coulomb forces when they interact with both the positively charged atomic core and the negatively charged electrons around the atoms most of the interaction occurs quite close to the atoms, within about one angstrom. Electron diffraction also plays a major role in the contrast of images in electron microscopes.Įlectron diffraction occurs due to elastic scattering, when there is no change in the energy of the electrons during their interactions with atoms. These patterns are similar to x-ray and neutron diffraction patterns, and are used to study the atomic structure of gases, liquids, surfaces and bulk solids. 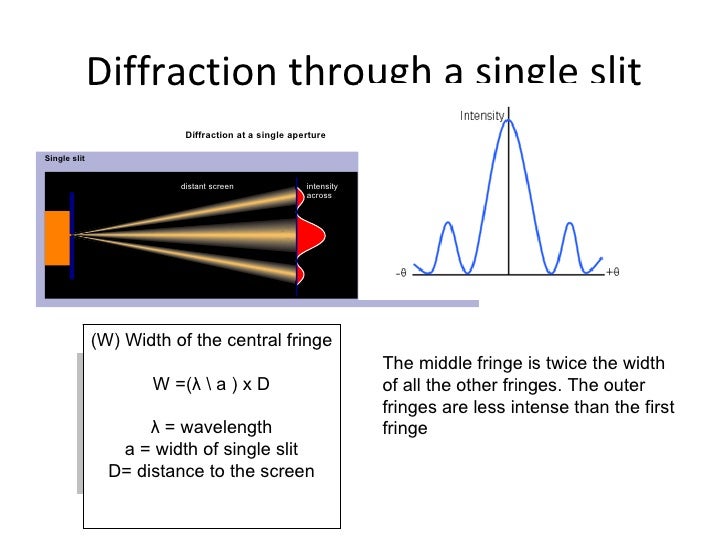
The resulting map of the directions of the electrons far from the sample ( Fraunhofer diffraction) is called a diffraction pattern, see for instance Figure 1. Close to the atoms the changes are described as Fresnel diffraction far away they are called Fraunhofer diffraction. Bending of electron beams due to electrostatic interactions with matter Figure 1: Selected area diffraction pattern of a twinned austenite crystal in a piece of steelĮlectron diffraction refers to changes in the direction of electron beams due to interactions with atoms.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
